
Molecular geometry, thanks to natural forces, seeks the lowest energy solutions to every bond, so some molecules with central atoms and four connected, surrounding atoms are not tetrahedral.\): Geometric structure of methyl isocyanate (CH 3–N=C=O), note there is no rotation around the double bonds only the single CN bond can rotate. The repulsion seeks its lowest energy level, providing the widest possible dispersal of the surrounding atoms. VSEPR theorizes that the lone pairs perform the same task as the bonds, repelling electrons to distribute joined atoms at equal angles around the central atom. For tetrahedral molecules like methane or xenon tetroxide, their steric number is four four bonds atom to atom and no lone electron pairs. All the bonds to the central atom, plus all the lone pairs, equals the molecule's steric number. Valence Shell Electron Pair Repulsion Theory, or VSEPR (pronounced "Vesper") predicts the molecular geometry of individual molecules. Lone pairs are the valence electrons of the atom that are not shared with another atom. Methane (CH4) (a) Structure C O O Double Bond H O Linear Molecular Geometry H 104.4o Bent Molecular Geometry C O Trigonal Molecular Geometry H H C Tetrahedral. The many shapes of molecules are affected by the number of atomic bonds and lone electron pairs. 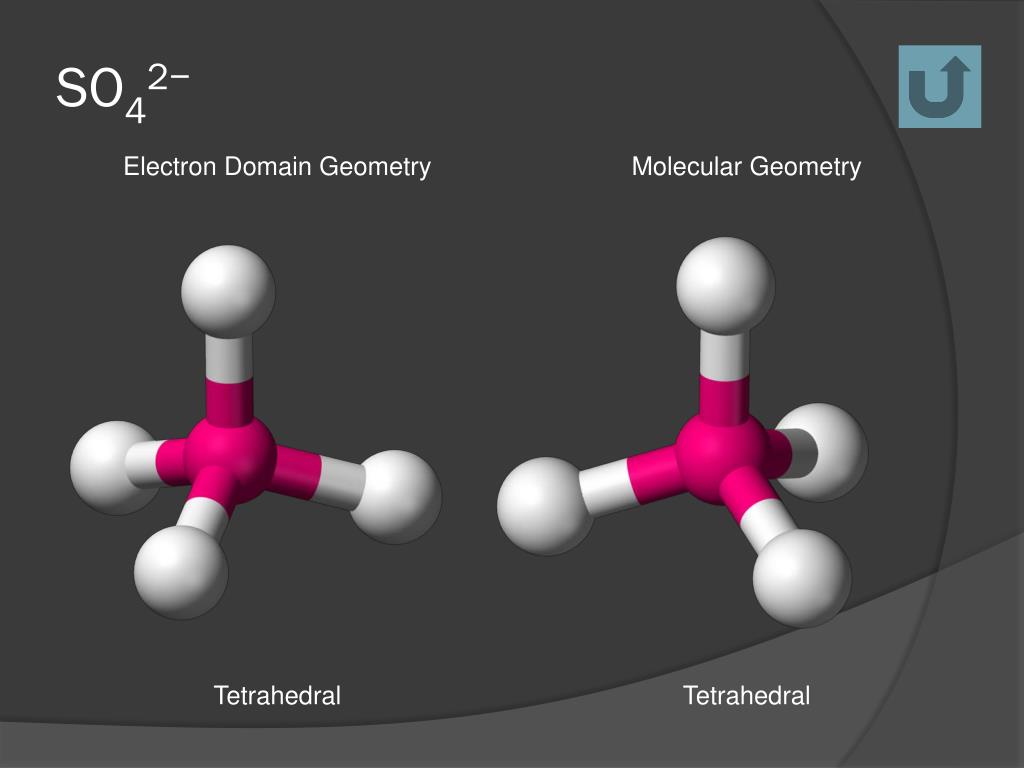
Valence shell electron pair repulsion theory (VSEPR) It pushes the molecule into a three-dimensional structure.Ĭhemists have worked hard to explain the actual structure of molecules, developing a theory connecting geometry, energy, and atoms. Many shapes exist beyond tetrahedrals, but we are concentrating on that shape here. We need to overcome this to obtain a stable Lewis structure for C 2 H 4. Tetrahedral - SP3 hybridized, like methane, CH4, with the hydrogen atoms arrayed around the carbon atom at 109.5° bond angles in three dimensions However, as can be seen above, the Carbon atom on the left only has 6 valence electrons in its grasp. Trigonal planar - SP2 hybridized, like sulfur trioxide, SO3, with the oxygen atoms 120° apart in one plane, the sulfur atom at their center Linear - SP hybridized, like CO2, with oxygen atoms 180° apart from one another, with the carbon atom between them The formal charge in the SiO2 lewis dot structure is zero. The total valence electron available for the Silicon dioxide lewis structure is 16. The bond angle of Silicon dioxide is 180º and the hybridization of it is Sp. We humble humans "discovered" geometry long after the universe had been putting it to use shaping the molecules that define our world.Īs chemical bonds form, individual atoms and their orbiting electrons move into specific shapes, called their electron domain geometry: The electron and molecular geometry of SiO2 are linear. Some might even say molecules developed geometry first. We see the structure of molecules in chemistry connecting to geometry in the field of molecular geometry. Down to the scale of molecules, geometry still holds. Geometry and real life are full of surprising alignments. Valence Shell Electron Pair Repulsion (VESPR) Theory attempts to explain the natural repelling forces of these electron arrangements. The total of bonds and lone pairs is a molecule's steric number. 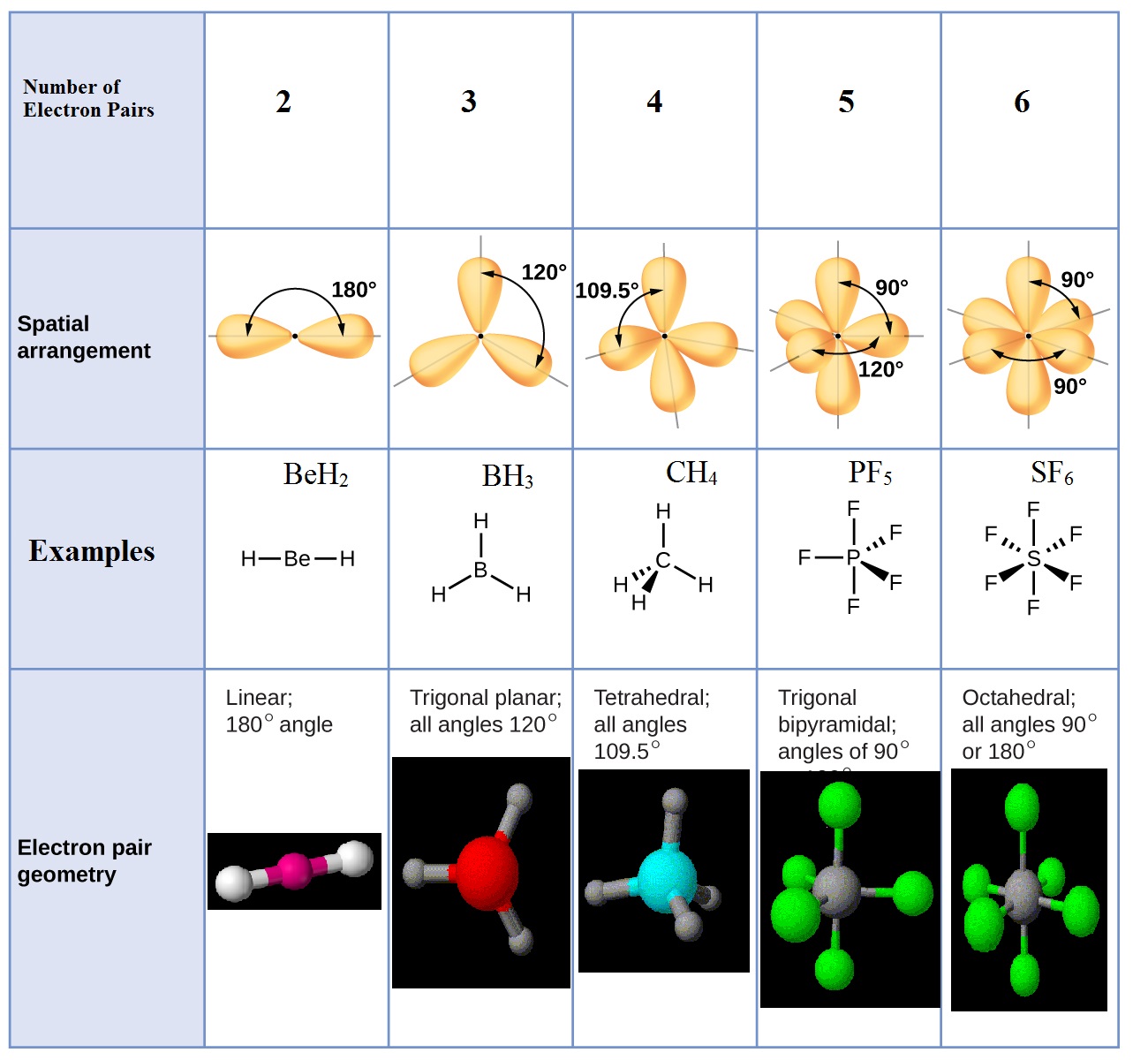
Molecules achieve their shapes from the atomic bonds and lone pairs of electrons. When one or more of the bonding pairs of electrons is replaced with a lone pair, the molecular geometry (actual. The total number of electron pairs, both bonding pairs and lone pairs, leads to what is called the electron domain geometry. Molecular geometry is the study of the physical shape of molecules. The molecular geometries of molecules change when the central atom has one or more lone pairs of electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
